|
By Jill Escher
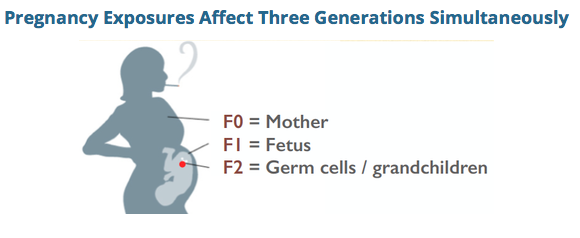
There is increasing appreciation that a subset of ASDs may be driven by heterogenous de novo errors in the germline (egg or sperm leading to the conceptus) or early conceptus, and also that epigenetic errors might be at play in some forms of autism. It seems reasonable to ask the next question, if there exist such molecular glitches in the germline, might they have been precipitated by certain mutagenic or epimutagenic exposures? As evidence mounts that autism is strongly heritable but not strongly genetic in the Mendellian sense, these questions appear to be increasingly reasonable. My work promoting gene-environment investigation in autism etiology is based in part on my own story. As an embryo and fetus was heavily exposed to a variety of synthetic steroid hormone drugs that were part of a popular, yet ineffective, "anti-miscarriage" protocol of the 1960s. I hypothesize these acute exposures induced subtle molecular errors, likely of an epigenetic nature, during reprogramming in my vulnerable early oocyctes, resulting in the delayed phenotypic consequence of my children's severe yet idiopathic dysregulated neurodevelopment (labeled as autism), decades after the exposure itself. I have found numerous autism families with strikingly similar evolutionarily novel exposures and strikingly similar unexplained pathologies in their offspring. But as I interviewed autism parents to investigate their prenatal histories, another exposure kept popping up. Over and over I would hear comments like: "No I don't think my mom took any drugs when she was pregnant with me, but she smoked like a chimney," or "My mother chain-smoked, so did my dad, and my mom kept smoking all the way through labor." At first I paid little attention to these anecdotes as I considered cigarette smoking to be mundane and harmless relative to the potent geno-affective chemicals to which I had been exposed. But in my interviews, stories of "grandma's smoking" and often grandpa's too, began to display an strong pattern, even though I started my interview project not remotely interested in this exposure. Grandmaternal cigarette smoking makes scientific sense in relation to de novo germline errors When one considers the well-established mutagenic effects of tobacco smoke, in combination with the critical developmental window of fetal germline synthesis, when the germline is stripped of methylation and dynamically remodeled, the biological plausibility of the hypothesis begins to add up. High toxicity. Tobacco smoke has many toxic and mutagenic components. There is also growing appreciation for its epimutagenic effects. Components of concern include BaP, nicotine, tar, formaldehyde, and benzene, not to mention additives and even pesticide and radiation residues. Research has shown that tobacco smoke and/or its constituents cause increased rates of mutation in gametes. DeMarini, in "Declaring the existence of human germ-cell mutagens," Environmental and Molecular Mutagenesis 2012, states that many studies have shown cigarette smoke to be a somatic-cell mutagen, and is genotoxic to oocytes and sperm of smokers. Marchetti et al, in "Sidestream tobacco smoke is a male germ cell mutagen," PNAS 2011, reflected that cigarette smoking increases oxidative damage, DNA adducts, DNA strand breaks, chromosomal aberrations, and heritable mutations in sperm. The study showed that short-term exposure to mainstream tobacco smoke or sidestream tobacco smoke (STS), the main component of second-hand smoke, induces mutations at an expanded simple tandem repeat locus (Ms6-hm) in mouse sperm. Passive exposure to cigarette smoke can cause tandem repeat mutations in sperm under conditions that may not induce genetic damage in somatic cells. Recent work by Yauk et al (unpublished) in a mouse model suggests increased rates of somatic mosaicism as a result of male germline exposure to benzo(a)pyrene, a mutagenic component of cigarette smoke, exposure. A recent study from the NIEHS, "DNA Methylation in Newborns and Maternal Smoking in Pregnancy: Genome-wide Consortium Meta-analysis," http://www.cell.com/ajhg/fulltext/S0002-9297%2816%2900070-7, found more than 6,000 differentially methylated CpG sites in newborns of mothers who smoked during pregnancy. Germline damage demonstrated. With respect to F1 paternal smoking, studies have shown reduction of quantity and quality of sperm in tobacco-using males, including a mouse study by Yauk finding changes in the DNA sequence of sperm cells. See Yauk et al, Mainstream tobacco smoke causes paternal germ-line DNA mutation, Cancer Res. 2007. Why examine early germline exposure instead of closer to the time of conception? As autism researchers increasing turn attention to gene-environment interactions in autism etiology, there have been efforts to seek "pre-conception" exposure data in certain cohorts. While this is a positive development, it ignores the biological reality that the most vulnerable period of germline development is the earliest phase (ie, in the embryo, fetus, and neonate) in which the dynamic rearrangement of germline epigenetic reprogramming, including the laying of imprints, occurs. As with somatic development, the germline, too, has "critical windows" in which vulnerability to exposures is more acute than in other phases. With respect to proximal exposure of the fetal brain, as opposed to germline, though fetal smoking exposure is associated with a wide variety of adverse outcomes and birth defects, there appears to be no evidence to support a measurable association between maternal prenatal smoking and ASD in offspring. See Rosen et al, "Maternal Smoking and Autism Spectrum Disorder: A Meta-analysis," Journal of Autism and Developmental Disorders 2015. However, germline exposure effects have not been probed. This could be a "smoking gun" In the big picture of potential mutagenic and epimutagenic agents, why focus on F0 cigarette smoking, instead of, say, synthetic sex steroid drugs or general anesthesia, which are two of the other potent prenatal exposures which repeatedly came up in the autism family interviews? Here are two reasons, aside from the known mutagenicity aspect: High prevalence. F0 maternal smoking appears to have been one of the most prevalent and acute toxic exposures during the timeframe in which the new generation of autism parents—and their germ cells—were in utero. Up to 40% of women smoked at the peak of the late 1960s. Furthermore, the exposure was often heavy—5,400 cigarettes through gestation for a pack-a-day smoker. Ease of ascertainment. It is easier to ascertain F0 smoking than use of other medications or medical interventions, simply because smoking would remain in the recollections of family members. (For example, I would not have known anything about my in utero exposures were it not for the exceptionally rare circumstance of having obtained detailed records of pharmaceuticals given to my mother while pregnant with me. My mother never told me about the medications, and did not smoke.) Let's get this research party started Sensing that high rates of maternal smoking in the second half of the 20th century may have unanticipated but lasting genomic consequences, Escher Fund for Autism seed funded a number of small pilot studies (for example, Denmark Prenatal Development Project cohort, CHDS cohort, UK ALSPAC cohort) that can begin to probe potential grandchild (F2) neurodevelopmental sequelae of grandmaternal (F0) smoking, via child (F1) prenatal smoking exposure. It will likely take years for pilot data to be sorted out. In addition, none of these projects are based on cohorts of autism subjects for whom we have genomic data. Escher Fund is also communicating with other researchers in an attempt to persuade them to examine F0 smoking as a variable in their autism cohorts, including: Australia Autism Biobank MSSNG (University of Toronto, Autism Speaks) Kaiser Autism Family Genetics Study (Kaiser) Korean Autism Cohort study (UCSF) African American autism cohort (UCLA) CHARGE study (UC Davis) Some observations from my interviews with autism families Based on my many interviews with autism families in which at least one biological grandmother had smoked heavily during pregnancy, I'd like to offer some preliminary observations: --The families had no prior history of autism --ASD cases where grandmother was a heavy smoker tend to be severe and nonverbal--perhaps there is a dose-response effect --The ASD is usually sporadic among siblings, but often siblings have other pathologies such as ADHD or learning disabilities --Sometimes F2 cousins descended from the same F0 grandmother who smoked have ASD --The grandfather was often a smoker too, meaning second-hand smoke could be a factor --There seem to be more multiplex F2 ASD children in African American families where a grandmother had smoked --Where F0 paternal grandmother had smoked, F1 father had smoked, and F1 father was of an older age, the risk for ASD in the F2 offspring seems to be markedly higher I hope more and more autism research can focus on this question of germline exposure to cigarette smoke, rather than continuing down the path of "genetics in a vacuum." My hunch, after conducting about 200 autism family interviews and reviewing many studies probing germline toxicity of cigarette smoke, is that heavy maternal smoking of the 1950s-70s could be the single greatest contributor to the heritably-driven surge in idiopathic abnormal neurodevelopment we see today.
1 Comment
Lisa Santana
2/19/2019 03:16:09 am
As a doctoral student, on hiatus from studies at this time, I found your article/blog interesting, well-written,and concise. My question are there any particular variables you have found within your studies to concur or/include how illegal drugs may also play a role on differential factors prior to pregnancy, and/or within DNA spectrum in exploring more research on Autism?
Reply
Your comment will be posted after it is approved.
Leave a Reply. |
AuthorJill Escher, Escher Fund for Autism, is a California-based science philanthropist and mother of two children with severe autism, focused on the question of how environmentally induced germline disruptions may be contributing to today's epidemics of neurodevelopmental impairment. You can read about her discovery of her intensive prenatal exposure to synthetic hormone drugs here. Jill is also president of Autism Society San Francisco Bay Area. Archives
July 2021
Categories |
- Home
-
Expert Q&A
- Eva Jablonka Q&A
- Amander Clark Q&A
- Mirella Meyer-Ficca Q&A
- Janine LaSalle Q&A
- Dana Dolinoy Q&A
- Ben Laufer Q&A
- Tracy Bale Q&A
- Susan Murphy Q&A
- Alycia Halladay Q&A
- Wendy Chung Q&A
- Pradeep Bhide Q&A
- Pat Hunt Q&A
- Yauk and Marchetti Q&A
- Emilie Rissman Q&A
- Carol Kwiatkowski Q&A
- Linda Birnbaum Q&A
- Virender Rehan Q&A
- Carlos Guerrero-Bosagna Q&A
- Randy Jirtle Q&A
- Jerry Heindel Q&A
- Cheryl Walker Q&A
- Eileen McLaughlin Q&A
- Carmen Marsit Q&A
- Marisa Bartolomei Q&A
- Christopher Gregg Q&A
- Andrea Baccarelli Q&A
- David Moore Q&A
- Patrick Allard Q&A
- Catherine Dulac Q&A
- Lucas Argueso Q&A
- Toshi Shioda Q&A
- Miklos Toth Q&A
- Piroska Szabo Q&A
- Reinisch Q&A
- Klebanoff Q&A
- Denis Noble Q&A
- Germline in the News
- Science
- Presentations
- About Us
- Blog
Proudly powered by Weebly

 RSS Feed
RSS Feed