Chemicals Can Exert Direct Epigenetic Effects on Exposed Fetal Germ Cells:
An interview with Piroska Szabó, PhD
"I am concerned about harming the exposed germ cells by the chemicals we
|
|
LINKS:
Recent Paper in Genome Biology: Iqbal K, Tran DA, Li AX, Warden C, Bai AY, Singh P et al. Deleterious effects of endocrine disruptors are corrected in the mammalian germline by epigenome reprogramming. Genome Biol. 2015; 16:59. Recent commentary in BioMed Central:
Does the germline remember? Van Andel Institute vai.org<http://vai.org |
You looked at three common environmental chemicals (the plasticizer Bisphenol A, the phthalate plasticizer DEHP, and the fungicide Vinclozolin) that are known to have “endocrine-disrupting” effects. Why did you choose these chemicals?
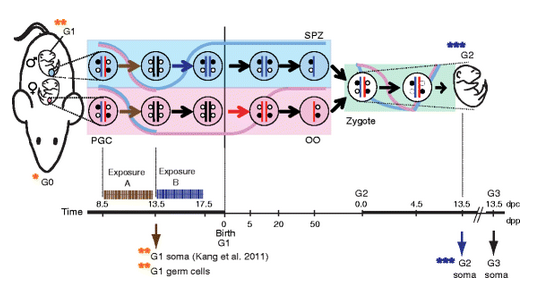
Vinclozolin has been reported to cause measurable phenotypic effects transgenerationally. The authors proposed that the phenotypic changes, including reduced fertility, were caused by altered DNA methylation in the sperm cells that were passed on between generations. This claim was very intriguing, instead of genetic inheritance of a mutation that inactivates a gene, the authors proposed the possibility of epigenetic inheritance of DNA methylation, which inactivates a gene by transcriptional repression. However, the hypothesis for this potential mechanism was based on a preliminary experiment and was not convincing. To understand the mechanism in greater detail we wanted to pinpoint the exact changes in the sperm genome and catch them at the time when they are triggered in the germ cells of the exposed fetuses. We wanted to follow the epigenetic effects of the chemicals between generations by mapping DNA methylation and RNA transcription at the genome-wide scale. In addition as control chemicals we included the anti-androgenic DEHP, which is similar to Vinclozolin, and BPA, which is an estrogen-like chemical. We chose these, because these are relatively well studied endocrine disrupting chemicals (EDCs), and each have been shown to cause morphological or physiological changes and induce differences in gene expression in mouse fetuses after treating the pregnant dams. We chose established and verified doses that are known to reach the fetus and our results confirmed that these chemicals have reached the germ cells of the fetus. Your data showed the male germline suffers immediate epigenetic effects after in utero exposure to BPA, Vinclozolin, and DEHP. Would you summarize the disruptions you detected in the germline? We measured genome-wide gene transcription changes in fetal germ cells that have been exposed inside the fetus and inside the mother’s womb. We compared the transcription levels of all protein coding genes in the exposed germ cells to the levels of control oil-carrier treated germ cells. When we used strict statistics for calling differences, we found that the gene transcription was affected slightly at a very small number of genes in the DEHP and VZ treated fetal male germ cells. When we used less strict statistics –to reduce the mistake of not finding a real hit (but on the down side, also increasing the risk of finding false positives), we found that male germ cells directly exposed to DEHP, VZ and BPA had more hits than male germ cells in the next generation that received no further treatment. This suggested that some of those hits were real positives. Gene pathway analysis revealed that the transcription changes have affected the reproductive or endocrine system in each case. We identified β -estradiol as the upstream regulator for BPA-treated MGCs, with a very high statistical probability; four transcripts of the top 10 changes are known to be regulated by β –estradiol. This was very interesting, because BPA is a β –estradiol mimicking chemical. In addition, hydroxysteroid (17-beta) dehydrogenase 3 (Hsd17b3) was upregulated by BPA and VZ, while androgen receptor (Ar) was upregulated by VZ and DEHP. These transcripts are interesting because they play roles in the male sex hormone pathways. Your project looked specifically at the molecular epigenomic mechanism of DNA methylation. How do artificial hormones or hormone mimics change DNA methylation in the early germ cell? What is it, structurally, about a chemical that makes it a hormone disruptor or hormone mimic? Does it lie in its affinity for hormone receptors that populate the cell nucleus?
Our study aimed to find such molecular changes in DNA methylation after exposure, but we found very small and insignificant changes in DNA methylation in fetal germ cells and also in sperm DNA. We are more confident about the changes we found at the level of gene transcription (please see above). These latter events are likely caused by epigenetic mechanisms other than DNA methylation. It is believed that artificial hormone-like chemicals have the potential to change gene transcription. One way of their action is based on their molecular structure and its similarity to natural hormones and the affinity of cellular hormone receptors to those structures. What other epigenomic layers might EDCs and hormone mimics affect? Histone modifications? If so, how? EDcs may affect gene transcription by causing changes in other epigenetic mechanisms, for example in histone covalent modifications or in non-coding RNAs. Histone proteins are very important, not only do they provide structural support to DNA in the form of nucleosomes, they also contain many different covalent modifications, which are, in turn are interpreted by the transcriptional machinery. Histone composition is known to be responsive to hormonal stimuli and is likely affected by EDCs. Future research will need to focus on the effects of EDCs on chromatin and especially on how they could affect histone modifications in the germline. Another layer of regulation occurs by long non-coding RNAs; they are also shown to have an effect on chromatin and transcription. The fetal period is a time of dynamic epigenetic remodeling of the germline. Why might this be a particularly vulnerable time for EDC exposures? The phenotype of the somatic cells (kidney, liver, brain, etc.) in our bodies is relatively stable. The long-term “lock-in” mechanism is provided by DNA methylation patterns that are maintained through many-many cell divisions. Embryonic germ cells, however, are stripped from DNA methylation. Histones, on the other hand stay on, even though they also undergo remodeling. Histone modifications are generally thought of as less stable, short-term epigenetic marks that are easier to be affected by different stimuli including hormones. It is logical to think that embryonic and fetal germ cells are more vulnerable to EDCs during the time when DNA methylation is temporarily absent. DNA methylation returns in the late fetal male germ cells and its pattern can be affected by the underlying histone patterns. If the histone remodeling is affected, one can expect a measurable change in transcription and perhaps also in DNA methylation. Can you say something about how imprinted genes differ from other genes in the human genome? And what, molecularly, is meant by the “imprints”? Imprinted genes are a small group of genes that exhibit parental allele-specific gene transcription in somatic cells. This means that even though there are two copies of each gene in each cell, only one is expressed; either in the chromosome that came from the sperm (paternally expressed) or in the other chromosome that came from the egg (maternally expressed). The cell can recognize a difference or “imprint” between the maternally and paternally inherited chromosomes and interpret these differences. In molecular terms, imprints are found in the form of a differentially methylated region (DMR), where methylation originates from the sperm or from the egg. Such DMRs control the parental allele-specific transcription in most imprinted domains, and these DMRs are then called imprinting control regions. Can you explain the timeframe for laying of imprints on genes in human gametes and how it differs between males and females? Imprints are valid for one generation, for the life of each individual. However, each person will pass on one kind of “imprint” in his/her gametes. Mothers will pass on maternally marked chromosomes in their oocytes whereas fathers will pass on paternally marked chromosomes in their sperm cells. The imprints switch their identity in the germlines. Embryonic primordial germ cells of both sexes erase the paternal and maternal imprints and following that the male germ cells establish paternal imprints during the fetal stages while female germ cells establish maternal imprints after birth in the juvenile growing oocytes. Are imprinted genes more or less protected during the process of epigenetic reprogramming? Are the periods of erasure, maintenance, and establishment distinctive in terms of environmental vulnerabilities? Based on this study alone it is not possible to say whether imprinted genes are more or less protected than the rest of the genome. We found that imprinted genes are very robust in their erasure, establishment and maintenance after exposing mouse germ cells at specific developmental stages in utero. When we specifically looked at the allele-specific imprinted gene expression or DNA methylation of a large set of imprinted genes, we detected no significant changes (after multiple statistical testing) in fetuses that derive from VZ, DEHP or BPA-exposed fetal male germ cells. We did not detect changes in the level of DNA methylation at imprinted genes in the exposed fetal male germ cells nor in the sperm of those male adults that had been exposed at the fetal stage. We also tested the effects of the same EDs on the erasure processes, and these also occurred without disturbance. With imprinting, it’s been said that subtle changes in the epigenome can cause outsize effects in terms of resulting pathologies. Do you think this is true?
According to other researchers, imprinted domains interact with other imprinted domains and form a so-called imprinted gene network. It has been reported that affecting one node of the network can affect another node. Those experiments involved drastic changes in one node when affecting another. Your question can be rephrased to mean, whether subtle changes in one imprinted node can affect other nodes inside the network and possibly downstream targets. In this case one would need to define what we consider subtle changes. I think that finding a one-to-five percent change of DNA methylation at an imprinted gene will not drastically change the phenotype even if the change appears highly significant in statistical terms. Indeed, there is already a variation between normal individuals in a population and often the methods of detection have a small rate of error. To answer your question one will need to search for pleiotropic effects after subtle disturbances at single imprinted loci. What do you think might be some broader implications of your research? From a scientific perspective, I believe that we have shown a considerable robustness of the epigenome remodeling processes in the germ line. This may even be important from an evolutionary point of view. Our study has also prompted an insightful debate in the literature. One interesting criticism we received was that studies aiming to disprove an effect should be more rigorously performed than those studies that claim to prove an effect. Apart from the fact that our intention was not to prove anyone wrong, I think that it will be an important take home message for every study to aim for the highest rigor and for clarity in the interpretation. It is our human nature that we want to find something positive, an effect, a correlation, a mechanism and the journals aim to publish positive results. Regardless, we should be objective when we interpret our data. We should be ready to accept our results and we should be clear and open about our limitations. I realize that your work did not replicate other labs' findings with regard to "transgenerational" effects of germ cell exposures. That means, effects in the subsequent, and unexposed, generation. But don't you think the fact that the F1 germline (F2 phenotype) suffers damage is in itself very significant from a public health point of view? Yes, you are right. I am concerned about harming the exposed germ cells by the chemicals we have tested. I also feel very concerned about potentially harming the germ cells by the many thousands of additional man-made chemicals that humans or wildlife can’t avoid being exposed to. The effects of those, on health or on the epigenome, not to mention the germ line, cannot all possibly be tested in a timely fashion. Chemicals that scientists prove harmful could be refused by consumers but can also easily be replaced by chemicals that are equally–or more–harmful, just think about BPA-free bottles that often contain BPA-like harmful chemicals. The effects of those chemicals may be amplified in mixtures with other chemicals, and they often come in mixtures. A lot of research will be needed to figure out those effects. It will continue to be important to measure whether the establishment of epigenetic patterns is disturbed in the germline and whether those aberrations are retained in the parental chromosomes of the offspring. What's next for your lab?
We are taking a step back from exposure research and study how DNA methylation patterns are determined in the germ cells. We will use genetic experiments to find the molecules and processes that are responsible for the normal patterns in fetal male germ cells. We showed earlier (Singh et al 2013) that DNA de novo DNA methylation takes place in male fetal germ cell only at sites where it is not excluded by methylation in the tail of H3 histone at Lysine 4 (H3K4me). Some enzymes are busy remodeling those H3K4me patterns just around the time when DNA methyltransferase enzymes establish de novo DNA methylation. We will test the role of those enzymes that build or remove H3K4me marks at specific sites. We also found that there is low-level broad transcription along the whole genome in male germ cells at these times. We will test, whether this is required in any way for the global DNA methylation establishment. We are also interested in figuring out how the methylation patterns are remodeled in the zygote, immediately following fertilization. The gametes (sperm and egg) are very highly methylated throughout the genome, but only a small portion of this DNA methylation remains in place. It is actually very important for normal development to keep that small portion of methylation in place. We are interested in the molecular mechanisms of the global removal of DNA methylation, and its maintenance at the specific locations. Again, histone modifications can affect these patterns. It is becoming more and more clear that the epigenetic state of the gametes has profound importance to the life and health of the resulting embryo. You and your readers may be interested in an upcoming scientific meeting we are working on now, because it is directly related to what we have been discussing about the importance of the germline epigenome. The inaugural “Frontiers in Reproductive Epigenetics” symposium will be held on May 12, 2017 in Grand Rapids, Michigan. This meeting is co-organized by researchers at the Center for Epigenetics at Van Andel Research Institute (VARI) and the Department of Obstetrics, Gynecology and Reproductive Biology, at Michigan State University (MSU). The theme this year will be “Epigenetics of maternal and paternal effects”. The symposium will focus on emerging knowledge and questions concerning parental effects. In such cases, it is the genotype of the mother or father, rather than the genotype of the individual, that determines the phenotype. These phenomena are now being extensively investigated at the molecular level, and the results point to specific epigenetic modifiers affecting the epigenome of the egg or the sperm. It is becoming clear that DNA methylation and histone covalent modification, among other epigenetic features need to be placed properly in the sperm and oocyte to make a healthy gamete. It is absolutely essential that the enzymes that carry out highly orchestrated remodeling in the developing gametes are working properly. We will hear about findings such as: oocyte-derived DNA methylation plays a major role in regulating trophoblast (placenta) development; defective histone methylation in sperm causes developmental failure of the embryo, and so on. The audience will realize that maternal and paternal effects may explain cases of infertility where the baby has no defect in his/her genes, but the defect can be found in the parents’ genome only. Genetic disturbances in the gamete’s epigenome can have long-lasting effect on the offspring and even on future generations. You can find more information here: http://www.frontiersinreproepi.org/. |